MS provides qualitative and quantitative information about the atomic and molecular composition of inorganic and organic materials.
Mass Spectrometry - Instrumentation
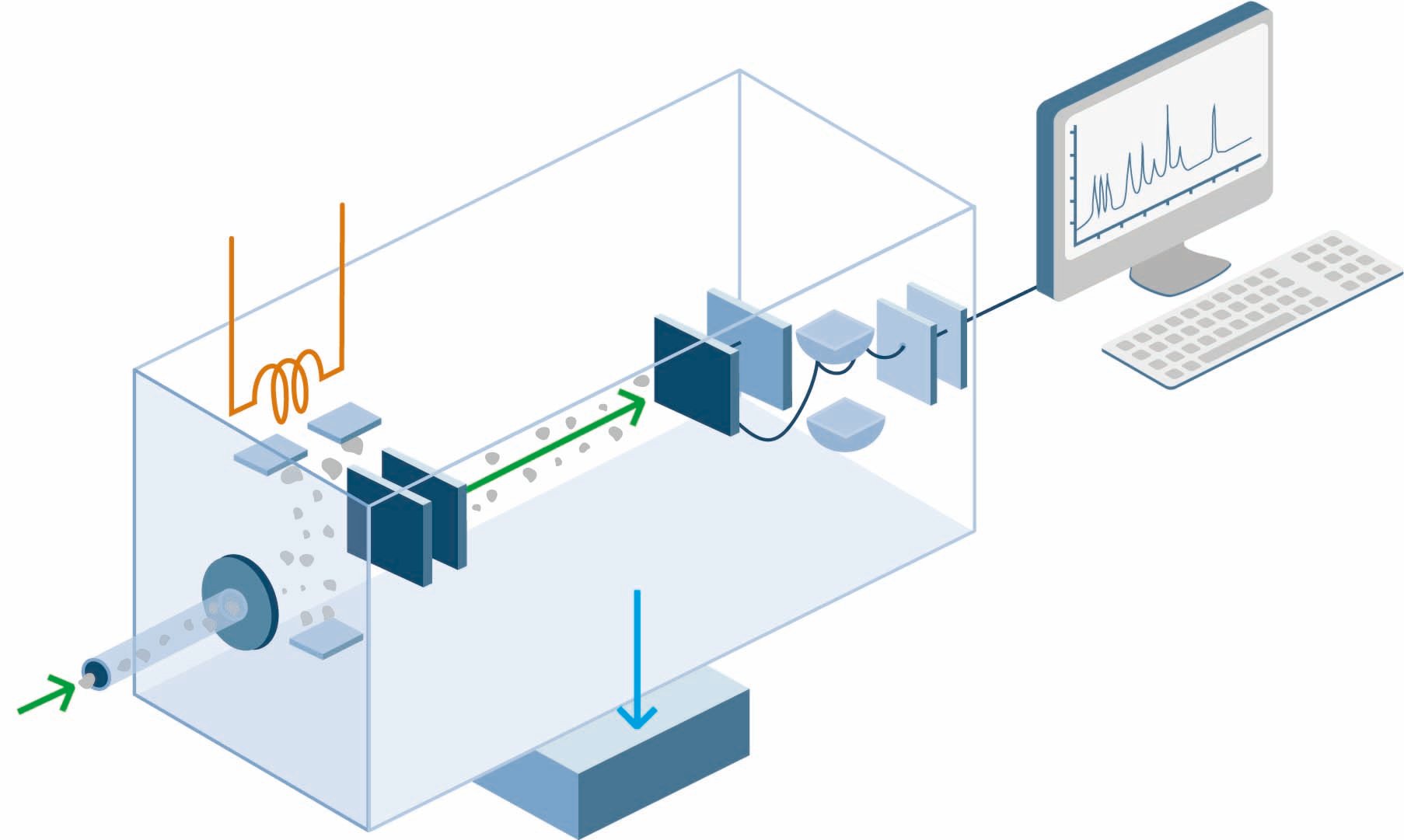
Mass spectrometers consist of four basic parts; a handling system to introduce the unknown sample into the equipment; an ion source, in which a beam of particles characteristic of the sample is produced; an analyzer that separates the particles according to mass; and a detector, in which the separated ion components are collected and characterized.
Detection limit |
Gas recommendation |
Regulator recommendation |
Gas generator recommendation |
| Calibration gas mixtures | |||
| ≤ 100 ppm | HiQ Calibration Mixtures | BASELINE C106 series | n/a |
| ≤ 100 ppb | HiQ Accredited Mixtures | REDLINE C200 series | n/a |
| ≤ 100 ppb | HiQ Calibration Mixtures | REDLINE C200 series | n/a |
| ≤ 100 ppb | HiQ Hydrocarbon Mixtures | REDLINE C200 series | n/a |
| Carrier gas (capillary column) | |||
| ≤ 100 ppm | HiQ Helium 5.0 | BASELINE C106 series | n/a |
| ≤ 100 ppm | HiQ Hydrogen 5.0 | BASELINE C106 series | HiQ PGH2 Hydrogen |
| ≤ 100 ppm | HiQ Nitrogen 5.0 | BASELINE C106 series | HiQ HPN2 Nitrogen |
| ≤ 100 ppb | HiQ Helium 6.0 | REDLINE C200 series | n/a |
| ≤ 100 ppb | HiQ Hydrogen 6.0 | REDLINE C200 series | HiQ NMH2 Hydrogen |
| ≤ 100 ppb | HiQ Nitrogen 6.0 | REDLINE C200 series | n/a |
| Collision cell gas | |||
| ≤ 100 ppb | HiQ Helium 6.0 | REDLINE C200 series | n/a |
| ≤ 100 ppb | HiQ Nitrogen 6.0 | REDLINE C200 series | n/a |
| Reagent gas (Chemical Ionisation) | |||
| ≤ 100 ppb | HiQ Ammonia 5.0 | REDLINE C200 series | n/a |
| ≤ 100 ppb | HiQ Carbon dioxide 4.5 | BASELINE C106 series | n/a |
| ≤ 100 ppb | HiQ Isobutane 3.5 | BASELINE C106 series | n/a |
| ≤ 100 ppb | HiQ Methane 5.5 | REDLINE C200 series | n/a |
Detection limit |
Gas recommendation |
Regulator recommendation |
| ≤ 1 ppb | HiQ Ammonia 5.0 | REDLINE C200 series |
| ≤ 1 ppb | HiQ Helium 6.0 | REDLINE C200 series |
| ≤ 1 ppb | HiQ Hydrogen 6.0 | REDLINE C200 series |
| ≤ 1 ppb | HiQ Argon 4.8 | BASELINE C106 series |
The spectrometer requires a collision free path for the ions and therefore operates under vacuum or near vacuum conditions. The sample inlet system is designed for minimal loss of vacuum. The ion source creates gaseous ion fragments from the sample. There exist two kinds of ion sources; gas-phase sources and desorption sources.
Gas-phase sources
In gas-phase sources, the sample is first volatilized before the ionization of the gaseous components takes place in various ways. The sample is vaporized outside the ion source. Examples of ionization methods are chemical ionization, electron-impact ionization and field ionization.
Chemical ionization
In chemical ionization, a small amount of gaseous atoms is ionized by collision with ions produced by electron bombardment of the reagent gas. Some widespread reagent gases are methane, oxygen, ammonia and hydrogen.
Electron-impact ionization
The electron-impact ion source is the most commonly used ionization method. An electron beam, generated from a tungsten or rhenium filament, is used to ionize gas-phase atoms or molecules. Ions are formed during collision of the electron beam and sample molecules.
M + e- -> M+. + 2e-
Here M represents the analyzed molecule and M+. is its molecular ion. The positive ions are accelerated by an electric field and passed into a magnetic field. By changing the accelerating voltage, i.e. the speed of the particle, or the magnetic field strength, ions of different mass-to-charge ratio can be collected and measured.
Field ionization
Molecules can lose an electron when placed in a very high electric field. High fields can be created in an ion source by applying a high voltage between a cathode and an anode, a so called field emitter. A field emitter consists of a wire covered with microscopic carbon dendrites, which greatly amplify the effective field of the carbon points. The gaseous sample from an inlet system is allowed in to the high-field area around the microtips of the emitter. Electrons from the analyte are extracted by the microtips and there is a little or no fragmentation of the ion.
Desorption sources
In desorption sources, ions are formed from samples in a condensed phase. A major advantage of desorption ionization is that it permits the examination of non-volatile and thermally unstable molecules. Two examples of desorption source are field desorption and fast atom bombardment.
Field desorption
Field desorption is a valuable technique for studying surface phenomena such as adsorbed species and the results of chemical reactions on surfaces. It is also a useful method for large lipophilic polar molecules. In field desorption a multi-tipped emitter similar to that employed in field ionization is used. The electrode is mounted on a probe that can be removed from the sample compartment and coated with a solution of the sample. Ionization takes place by the application of a high potential to the electrode. Sometimes it is necessary to heat the emitter with an electric current.
Fast atom bombardment
In fast atom bombardment, a high-energy beam of neutral atoms, typically xenon or argon, strikes a solid sample causing desorption and ionization. This technique is used for large biological molecules that are difficult to get into the gas phase. The atomic beam is produced by accelerating ions from an ion source through a charge-exchange cell. The ions pick up an electron in collision with neutral atoms to form a beam of high energy atoms.
Mass analyzer designs
The purpose of the mass analyzer is to separate the ions produced in the ion source according to their different mass-to-charge ratio. The most common analyzer designs include the quadruple, magnetic sector and time-of-flight mass analyzers.
Quadruple
A quadruple field is formed by four electrically conducting parallel rods. The applied voltages affect the trajectory of ions travelling down the flight path centred between the four rods. For given voltages, only ions of a certain mass-to-charge ratio are allowed to pass through the quadruple filter, while others are carried away as uncharged molecules. By varying the electrical signals to a quadruple it is possible to vary the range of the mass-to-charge ratio transmitted. This makes spectral scanning possible.
Magnetic sector analyzer
The magnetic sector analyzer employs a magnetic field that causes ions to travel in a circular path of 180, 90 or 60 degrees. Initially ions are accelerated through slit B into the metal analyzer tube. Ions of different mass can be scanned across the exit slit by varying the field strength of the magnet or the accelerating potential between slit A and B. The ions passing through the exit slit fall on a collector electrode, resulting in an ion current that is amplified and recorded.
Time-of-flight analyzer
A time-of-flight mass spectrometer uses the differences in transit time through a drift region to separate ions of different masses. The ions from the ion source are accelerated by an electric field pulse. The accelerated particles then pass into a field-free drift tube that is about a meter in length.
The essential principle of time-of-flight mass spectrometry is that all ions are accelerated to the same kinetic energy. Their velocities are inversely proportional to the square roots of their masses. The lighter ions of high velocity arrive at the detector earlier than the heavier ions of low velocity.
Ion collection system
The ion collection system is measures the relative abundance of ion fragments of each mass. Several types of detectors are available for mass spectrometers. The detector used for most routine experiments is the electron multiplier. Another type of detector is photographic plates coated with a silver bromide emulsion, it is sensitive to energetic ions. A photographic plate can give a higher resolution than an electrical detector.